In Clinical
Follow this topic
Bookmark
Record learning outcomes
When the National Review of Asthma Deaths (NRAD) looked at 195 deaths caused by asthma in 20142, it identified one or more avoidable factors in 130 (67 per cent) of the cases.
It also found that:
- 27 (10 per cent) had no evidence in their records confirming they had asthma
- Overall asthma management was satisfactory in only 31 of the 195 people who died (and only one of the 28 children and young people).
The panel also identified a number of missed opportunities by healthcare professionals to intervene and reduce the risk of asthma attacks and death. These included:
- The overprescribing of SABAs (short-acting beta agonists) and insufficient provision of inhaled corticosteroid (ICS) preventer medication
- Failure to monitor asthma control and to provide follow-up assessment and optimisation of medication after asthma attacks
- Failure to refer patients to an asthma specialist
- The non-implementation of UK BTS/SIGN asthma guidelines in 89 of the deaths.
Sadly, nearly a decade later, many of these findings still hold true.
Over-reliance on SABAs
The UK has one of the highest uses of SABAs in Europe among people with asthma – about 38 per cent of asthma patients are using three or more SABA inhalers a year3 and these patients account for 83 per cent of the SABA inhalers prescribed.4
In England, there has been a modest improvement in patients using six or more SABA inhalers a year since the NRAD report but there is still a long way to go. EPACT 2 data from the NHS Business Services Authority in March 2016 showed nearly 30 per cent of asthma patients were receiving six or more SABAs over a 12-month period. This is now down to 23 per cent.
According to national guidelines, a person with good asthma control shouldn’t need to use their blue (SABA) emergency reliever inhaler more than twice a week. One blue inhaler containing 200 puffs should last at least six months.
With so many new therapeutic options available to help tackle current prescribing challenges, let’s now consider how pharmacy teams can intervene to:
- Help patients take the right preventative treatment at the right time to tie in with symptoms and reduce the risk of asthma attacks
- Help to improve medicines adherence and inhaler technique using SMART technology
- Identify those patients who may be suited to biologic treatments.
Women and asthma
Asthma + Lung UK has highlighted the fact that asthma is influenced by rising female sex hormones during puberty and then fluctuating levels during monthly menstruation cycles.
For many women their asthma can deteriorate significantly during the premenstrual period, with the most significant symptoms that worsen during this time being shortness of breath, wheezing and chest tightness.
Female hormones can also trigger asthma symptoms during the perimenopause (the time leading up to the menopause) –
a phenomenon known as perimenstrual asthma or PMA.
Of women with PMA, up to 68 per cent have previously been hospitalised for their asthma. They also report increased use of oral corticosteroids and increased healthcare utilisation compared to women without PMA.
While there may be little that can be done to reduce the sex/gender-linked risk, pharmacy teams can advise women who live with asthma about the extra importance of regular preventative treatment and minimising exposure to other triggers wherever possible.
Taking the right preventative treatment
Currently UK guidance recommends using two separate inhalers in mild and moderate disease – a preventer (ICS or ICS/LABA [long-acting beta agonist] and reliever (SABA) except in MART (Maintenance and Reliever Therapy) regimens.
One way of ensuring the preventer is taken when it is needed is to potentially combine an ICS with formoterol (a fast-acting LABA), which has properties like the SABA emergency relievers, eliminating the need for a separate blue inhaler.
The MHRA has approved the use of a dual (ICS/formoterol) combination treatment to be used as a reliever therapy in those aged 12 years and over. This therapy choice is situated early in the asthma treatment pathway as an alternative to its current use as a fixed dose preventer (or as MART therapy, which sits later in traditional treatment pathways).
The MHRA approval is currently for a budesonide 200mcg/formoterol 6mcg combination that is delivered as dry powder via a Turbohaler.
A potential advantage is that it avoids patients having to take unnecessarily high doses of ICS or ICS/LABA in the long-term and may reflect what patients with mild asthma naturally do (i.e. only use the inhaler when they feel symptomatic).
The reliever acts as the vehicle to deliver extra ICS at a time when it is most needed.
In recent trials, the use of this dual therapy, utilising the fast-acting property of formoterol in combination with ICS, resulted in a 51 per cent reduction in asthma attacks compared to the use of short-acting beta agonists alone and a 56 per cent reduction in severe asthma attacks compared to ICS daily treatment and SABA.5
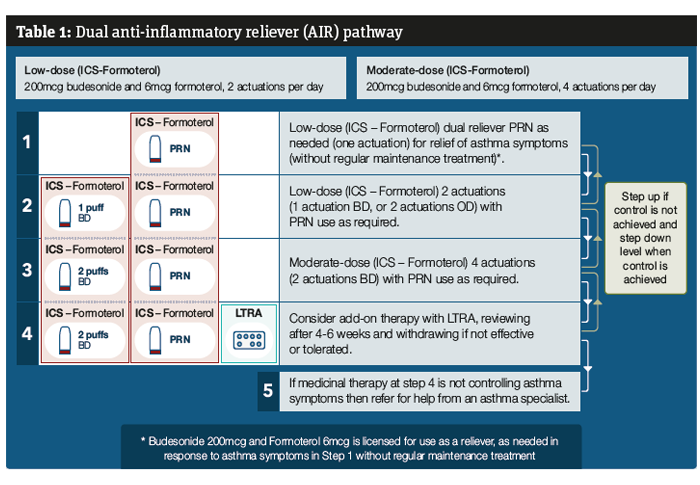
This new AIR (anti-inflammatory reliever) therapy option does not yet sit within an approved national guideline as NICE and BTS SIGN last published their treatment pathways in 2017 and 2019 respectively. The Primary Care Respiratory Society (PCRS) has looked to the latest Global Initiative for Asthma (GINA) approach to asthma treatment to see how this new approach fits and provided some guidance (see Table 1).6
What can pharmacy teams do?
Pharmacists can check which asthma patients may be suited to AIR or MART on the PMR. Look for:
- ≥6 SABA inhalers in the past 12 months or ≥3 in the past six months
- ≤3 or 5 ICS pick-ups a year
- Assessing courses of oral corticosteroids for asthma in the last year.
Utilising SMART technology
SMART technology may help to improve ICS adherence in those patients who need regular preventative treatment and potentially alert the patient to any inhaler technique errors.
Average adherence among patients using preventer medicines is between 22-63 per cent. This is poor considering preventers will help keep symptoms at bay and lower future risk of attacks.7
The reasons for this are complex and need further exploration. Difficult to manage asthma is a term often used by clinicians. Patients with difficult to manage asthma (i.e. those who have daily symptoms and regular attacks despite apparently best treatment) fall into two groups:
- People whose asthma has been controlled in the past but who have now lost control
- People whose asthma has never been controlled.
Investigations and management should aim to:
- Identify when asthma control is lost/has never been achieved, by prompt and effective monitoring
- Gain/regain control and then maintain control of asthma with effective, well tolerated treatment.
The Asthma Control Test (ACT) can help identify when control is lost as it assesses recent symptoms. Another useful indicator is to assess lung function e.g. when peak flow readings fall below 80 per cent of their best, which should be cross checked against the patient’s latest asthma plan. SABA and oral corticosteroid over-reliance and ICS underuse can be picked up from GP records/PMRs in community pharmacy. Inhaler technique errors can be picked up through NMS interventions or during routine asthma reviews.
Clinicians can check for both unintentional and intentional non-adherence by asking the patient how they take their medication.
The NHS Long Term Plan in 2019 advocated that SMART inhalers would be piloted so patients can easily monitor their condition.8
Recent developments in the last few years include Teva Pharmaceuticals launching an all-in-one digital inhaler (GoResp Digihaler; pictured bottom left) that could be prescribed. This can pick up some errors in inhaler technique (e.g. insufficient inhalation manoeuvre or multiple doses) and can monitor adherence via a companion mobile app.9 It is only licensed in people 18 years and over.
Enerzair Breezhaler, meanwhile, is an example of where a sensor has been co-packaged with the inhaler device to help patients with uncontrolled asthma.10 Again, this is only licensed for those aged 18 years and over.
Other examples include a Rafi-Tone app for children, which works alongside the Able Spacer and Positive Whistle Mask. When a good seal and adequate flow of breath is achieved, a whistle sounds. The app listens for a specific sound, using sound response technology and operates a fun and engaging game in response.
Smart Respiratory has sensors that can be added to inhalers that have no dose counters to help patients know when inhalers are empty or if they are overusing SABA or underusing ICS.
What can pharmacy teams do?
Refer those patients identified as having:
- Poor inhaler technique
- Infrequent ICS collection
- Poor knowledge of asthma or not taking the condition seriously
- Struggling to afford the cost of their prescriptions.
Biologic treatments
Asthma UK research found that three out of four people with suspected severe asthma in England were not being referred when they should be for receiving biologics.
Asthma biologics work in a targeted way by disrupting pathways causing airways inflammation, helping to manage symptoms, reduce relapses and reduce reliance on other medicines such as oral steroids.
A small cohort of patients (around 200,000 people – 5 per cent of the asthma population) will have severe asthma in which conventional treatments do not adequately control symptoms and lung function. Clinicians find the HASTE tool very useful.11
The good news is there are now potentially life-changing biologic treatments (monoclonal antibodies) available on the NHS. To date NICE has approved these biologics for severe asthma: omalizumab, mepolizumab, reslizumab, benralizumab and dupilumab. They are given as an injection or infusion (depending on which biologic is used), either regularly in hospital or self-injected by patients at home.12 They can reduce the number of asthma attacks and even reduce or stop the need for oral corticosteroids.13, 14
What can pharmacy teams do?
- Help patients understand the potential benefits of oral corticosteroids and also the harms they can cause if repeated doses are taken
- Identify patients suspected of having difficult to control asthma (some may have severe asthma) and refer for urgent review.